Quantitative Mass Spectrometry at IMSERC
Quantitation of Drugs in Biological Samples
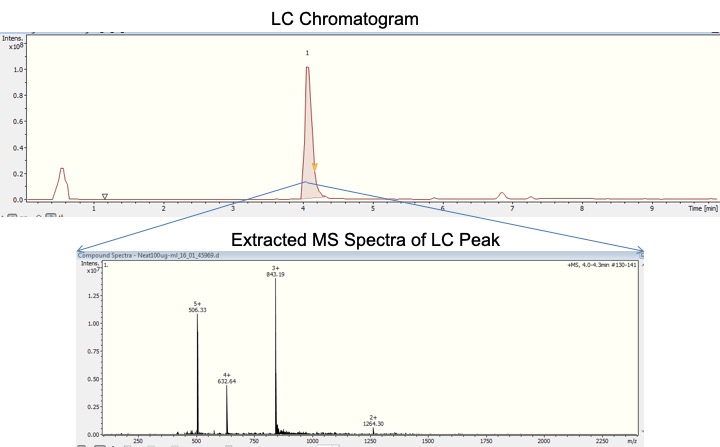
Liquid chromatography-mass spectrometry (LC-MS) is a combination of a High-Performance Liquid Chromatography (HPLC) system and a Mass Spectrometer (MS) that allows for the isolation and quantitative measurement of the analytes of interest in highly complex mixtures. LC separates compounds by their physicochemical properties, and MS differentiates ionized compounds by mass-to-charge ratio. Thus, the mass spectrometer acts not only as the LC detector, but it also helps to identify the species corresponding to each chromatographic peak through its unique mass spectrum.
These two techniques used in tandem provide a unique capability for highly sensitive and cost-effective quantitative determination of organic molecules in a variety of applications, including analysis of trace organic compounds in environmental, food, and biological matrices. Combining the two separation mechanisms of LC and MS allows particular analytes to be isolated from the mixture and gives confidence that the correct component is being measured. In addition, the technique allows for the use of isotopically labelled internal standards, which do not separate by LC from the analyte of interest, but these standards can be separated by their mass difference. The use of stable isotopically labelled (SIL) internal standards help control variability in a quantitative assay.
IMSERC is equipped with walkup quantitative capabilities to enable any researcher to gain easily quantitative results. IMSERC primarily utilizes a SCIEX 6500+ QQQ or a Bruker AmaZon X Ion Trap for quantitative experiments using MS and/or MS/MS. The resulting selectivity of such quantitative techniques is demonstrated below.
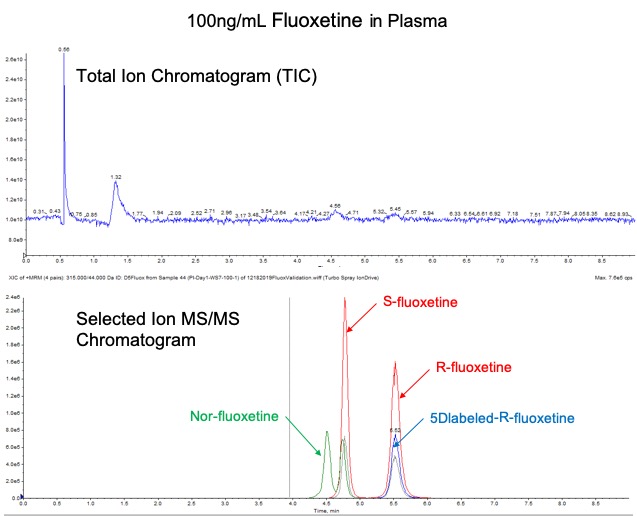
In the above graphic, (TOP) when using just a single stage of MS detection, it is oftentimes impossible to distinguish ion signals of interest from the total ion chromatogram (TIC); however (BOTTOM) when using MS/MS quantitative techniques to monitor specific fragmentation patterns of the analytes of interest, low levels of detection can be reached in the most complicated of matrices. In both of examples shown above, separation and quantitation of chiral isomers of the antidepressant drug, Fluoxetine, and metabolites detected in blood are demonstrated.
Also demonstrates above are the speed and specificity benefits gained in quantitation via LC/MS. The MS will distinguish compounds based on their unique mass or further using fragments created via tandem MS (or MS/MS or mass transitions). Therefore, the LC method does not have to separate every single component in the sample, as is necessary with UV detection, allowing for co-elution of analytes using a shorter method. This allows fast LC analysis times, reduced sample preparation, and high throughput sample analysis.